Buoyancy and phase changes in water
The page has moved; please visit below:

The page has moved; please visit below:

The page has moved; please visit below: Understanding overshooting top in thunderstorms

The page has moved; please visit below: Back-sheared anvil: The role of wind shear in shaping cumulonimbus anvil

The page has moved; please visit below: The beauty and mistery of mammatus clouds

The page has moved; please visit below: Types of convective clouds – from fair-weather clouds to thunderstorms

The page has moved; please visit below: Spotting convective weather: clouds and thunderstorms
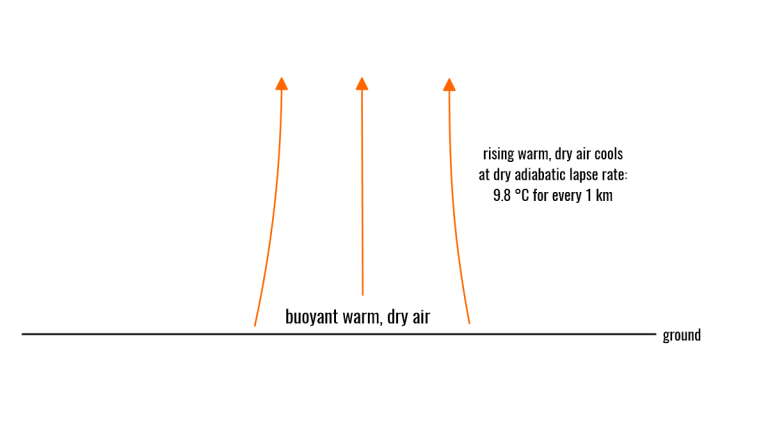
The page has moved; please visit below: The Science Behind Convection: How Does it Work?